Phytochemistry, In-vitro antioxidant, Microbicidal, Anti-ulcerogenic and Biosafety Potential of Emilia coccinea Aqueous Extract in Animal models.
Main Article Content
Abstract
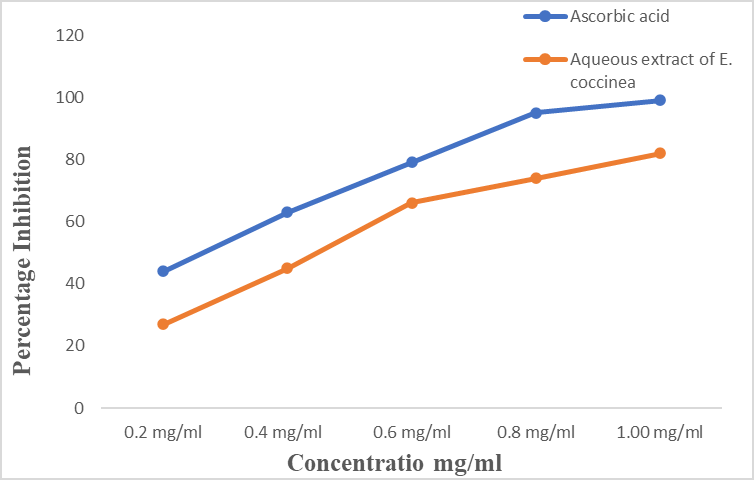
Emilia coccinea enhances the treatment of several disease conditions include; vertigo management, ringworm, cough, gonorrhoea, ulcers, lice, measles, seizure, eye drop. This study investigates the phytochemistry, In-vitro antioxidant, anti-ulcerogenic, microbicidal and biosafety effect of aqueous extracts of E. coccinea using animal model. E. coccinea leaf was freshly obtained, shade dried, pulverized and prepared into aqueous extract. Standard procedure were used for the evaluation of the phytochemicals, in-vitro antioxidant and antimicrobial activities. Twenty five (25) Wistar rats were acclimitized and randomly selected into five groups (n=5) such as untreated group, 10 mg/kg cimetidine and graded doses (100, 200 and 400 mg/kg b.w.) of aqueous extract of E. coccinea to evaluate antiulcer and biosafety effects. Results from the qualitative and quantitative phytochemical screening showed the present of phenol (47.19 mg), saponins (84.64 mg), alkaloids (75.17 mg), cardiac glycosides (63.12 mg) and anthraquinone (48.79 mg). The antioxidant property showed a competitive scavenging effect against DPPH radical when compared with ascorbic acids. Microbial activities of the aqueous extract of E. coccinea at various concentration elicited inhibitory effect against Salmonella typhii, Escherichia coli, Helicobacter pyloris. The ulceration in rat stomach lining induced with 70% ethanol and pretreated with prophylactic measure of aqueous crude extract showed significant reduction in ulcer count, ulcer index with increased % inhibition of ulcer when compared with untreated and reference control. The haematological and histopathological study shows no significant difference in the aqueous crude extract with absent toxicity when comparisons with the control.
Article Details
References
Wainright, M. Miracle cure: The story of penicillian and golden age of Blackwell. Publishers. London. 2001, 237p.
Kokate, C. K., Purohit, A. P., Gokhale, S. B. Pharmacognosy. Nirali Prakashan, Pune. 2006, 15-38pp.
Ozumba, U.C. Antibiotic sensitivity of Isolates of Pseudomonas aenigenosa in Enugu, Nigeria. African Journal Clinical and Experimental Microbiology. 2003, 44:48-51. DOI: https://doi.org/10.4314/ajcem.v4i1.7323
Olorode, O. Taxonomy of West African Flowering Plants. London Longman Publishing Company. 1984, 121.
Sofowora, E. A. Medicinal plants and traditional medicine in Africa. John Wiley and sons Chiclester. 1982, 198p.
Ayitey-Smith, E. Prospects and Scope, Plant Medicin in Health Care. Ghana University Press. 1989, 29p.
Sofowora, E.A. Medicinal plants and traditional medicine in Africa. Spectrum Boot Ltd. Ibadan, Nigeria. 1993, 289p.
Ogbebor, N., Adekunle, A.T. Ingibition of conidial germination and mycelia growth of Corynespora caiicola (Berk and Curt) of rubber (Hevea brasiliensis muell. Arg.) Using extracts of some plants. African Journal of Biotechnology. 2005, 4:996-1000.
Ndip, R. N., Tarkang A. E. M., Mbulla, S. M., Luma, H. N., Malenque, A. In vitro Anti- helicobacter phylori activity of extract of selected medicinal plants from North West Cameroon. 2007, 245p DOI: https://doi.org/10.1016/j.jep.2007.08.037
Edeoga, H. O., Okwu, D.E., Mbaebie, B.O. Phytochemical constituent of some Nigeria medicinal plants. African Journal Biotechnology. 2005, 4(7): 685-688. DOI: https://doi.org/10.5897/AJB2005.000-3127
Hill, A.F. Economic Botany. A textbook of useful plants and plant products. 2nd edn. McGarw-Hill Book Company Inc, New York. 1952.
Oliver, B. Medicinal Plants in Nigeria. Nigeria College of Arts, Science and Technology. 1960, p138.
Odugbemi, T. Outlines and pictures of Medicinal plants from Nigeria. University of Lagos press, Nigeria. 2006, 283-285.
Kerharo, J., Bouquet, A. Plants medicinales et toxiques de la Cote-d’ ivoire-Haute- Volta. Vigot Freres Paris. 1950, 295.
Milosavljevic, T., Kostić-Milosavljević, M., Jovanović, I., Krstić, M. Complications of peptic ulcer disease. Digestive diseases (Basel, Switzerland). 2011, 29(5): 491–3. DOI: https://doi.org/10.1159/000331517
Najm, W. I. Peptic ulcer disease. Primary care. 2011, 38(3): 383–94, DOI: https://doi.org/10.1016/j.pop.2011.05.001
Steinberg, K. P. Stress-related mucosal disease in the critically ill patient: risk factors and strategies to prevent stress-related bleeding in the intensive care unit. Critical Care Medicine. 2002, 30(6): 362–364. DOI: https://doi.org/10.1097/00003246-200206001-00005
Harborne, J. B. Phytochemical methods. London Chapman and Hall, Ltd. 1973, 49-188.
Ellof, J. N. A sensitive and quick microplate method to determine the minimum inhibitory concentration of plants extracts for bacteria. Planta Medica. 2008, 64: 711-713. DOI: https://doi.org/10.1055/s-2006-957563
Emeruwa, K. C. Antimicrobial substances from Carica papaya fruit extracts. Journal of Natural Products. 2009, 45(2): 123-127. DOI: https://doi.org/10.1021/np50020a002
Lorke, D. A new approach to practical acute toxicity testing. Archives of Toxicology. 1983, 54: 275-287. DOI: https://doi.org/10.1007/BF01234480
Karau D., Aly, S., Antonella, C., Carla, M., Jacques, S., Vittorio, C., Alfred, S.T. Antibacterial activity of alkaloids from Sida acuta. African Journal of Biotechnology. 2005, 4(12):1452-1457.
Sies, H. Oxidative stress: Oxidant and anti-oxidant. Experimental Physiology. 1997, 82(2): 291–295. DOI: https://doi.org/10.1113/expphysiol.1997.sp004024
Cadenas, E. Basic mechanisms of antioxidant activity. Biofactors. 1997, 6: 391–397. DOI: https://doi.org/10.1002/biof.5520060404
Oyewole, I. O., Magaji, Z. J., Awoyinka, O. A. Biochemical and toxicological studies of aqueous extract of Tithonia diversifolia leaves in Wister albino rats. Journal of Medicinal Plants Research. 2007, 1: 30-33.
O’Hara, M. A., Kiefer, D., Farrell, K., Kemper, K. A review of 12 commonly used medicinal herbs. Archive of Family Medicine. 1998, 7: 523-536. DOI: https://doi.org/10.1001/archfami.7.6.523
Idu, M., Omogbai, E. K. I., Aghimien G. E. I., Amaechina, F., Timothy, O., Omonigho S. E. Preliminary phytochemistry, antimicrobial properties and acute toxicity ofStachytarpheta jamaicensis (L.) Vahl. Leaves. Trends in Medical Research. 2007, 2:193- 198. DOI: https://doi.org/10.3923/tmr.2007.193.198
Ogbebor, N., Adekunle, A. T. Inhibition of conidial germination and mycelia growth of corynespoa casiicola (Berk and Curt) of rubber (Hevea brasilieusis Muella Arg). Using extract of some plants. African Journal Biotechnology. 2005, 4:996-1000.
Agrawal, N. M., Dajani, E. Z. Prevention and treatment of ulcers induced by non-sterioddal anti-inflammatory drugs. Journal on Gastroenterology. 1993, 3(4):142-148.
Curtis, W. D., Griffin, J. W. Non-steroidal anti-inflammatory drug induced gastroduodenal injury: therapeutic recommendations. Journal on Medical College Georgia Augusta Ailment Pharmarcology. 1991, 1:99-109. DOI: https://doi.org/10.1111/j.1365-2036.1991.tb00753.x
Cho, C. H., Ogle, C. W. The Pharmacological differences and similarities between stress and ethanol-induced mucosal damage. Life Sciences. 1992, 51: 1833-1842. DOI: https://doi.org/10.1016/0024-3205(92)90034-M
Schneeweiss, S; Maclure, M; Dormuth, C. R; Glynn, R. J; Canning C., Avorn, Journal Clinical pharmacology & therapeutics. 2006, 79(4): 379-386. DOI: https://doi.org/10.1016/j.clpt.2005.12.304
Konturek, P. C. H., Duda, A., Brzozowski, T. Activation of genes for superoxide dismutase, interleukin-1ß), tumour necrosis factor-α) and intercellular adhesion molecule-1 during healing of ischaemia-reperfusion gastric injury. Scand. J. Gastroenterol. 2000, 35: 452-463. DOI: https://doi.org/10.1080/003655200750023697
Adedapo, A.A., Abatan, M.O., Olorunsogo, O. O. Toxic effects of some plants in the genus Euphorbiceae on haematological and biochemical parameters of rats. Veterinarski Arhiv. 2004, 74(1):53–62.
Okonkwo, J.E., Iyadi, K.C., Effiong, C.O. Effect of chronic administration of haematological parameters of rats. Nig J Physiol Sc. 2004, 19(1–2):10–13
Yakubu, M.T., Akanji, M.A., Oladiji, A.T. Hematological evaluation in male albino rats following chronic administration of aqueous extract of Fadogia agretis stem. 2007, pp. 34–38.
Omodamiro, O.D., Nwankwo, C.I. The effect of Voacanga africana leaves extract on serum lipid profile and haematological parameters on albino wistar rats. European Journal of Experimental Biology. 2013, 3(3):140-148.