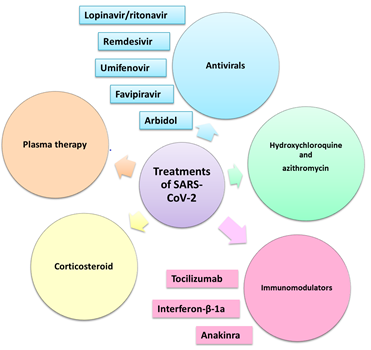
The main treatments used for SARS-CoV-2 patients
Main Article Content
Abstract
As the COVID-19 pandemic continues to ravage the world and threaten people's lives, treating infected patients effectively has been one of the top concerns for medical workers around the world. Currently, no medication is recommended to treat COVID-19, and no cure is available. Researchers are testing a variety of possible treatments. Several drugs are being researched in different countries. Most are existing drugs that are being trialled against the virus. Pharmaceuticals undergoing clinical trials to assess their safety and efficacy as potential treatments for COVID-19, include the antiviral nucleotide analogue remdesivir, systemic interferons and in particular interferon β-1a, the antiviral combination lopinavir/ritonavir, the antimalarial chloroquine/hydroxychloroquine, and monoclonal antibodies against components of the immune system such as interleukin-6 (IL-6) and IL-4. It is important that the potential treatments are carefully assessed in randomised controlled trials..
Article Details
References
Amanat F, Krammer F. SARS-CoV-2 vaccines: status report. Immunity, 2020; 52(4), 583-589. https://doi.org/10.1016/j.immuni. 2020.03.007 DOI: https://doi.org/10.1016/j.immuni.2020.03.007
Khan N, Fahad S. Critical review of the present situation of corona virus in China. Available at SSRN. 2020; 3543177. DOI: https://doi.org/10.2139/ssrn.3543177
Memberships M, Join T. Guidance on Coronavirus Disease 2019 (COVID-19) for Transplant Clinicians Updated 27 February 2020.
World Health Organization (a). Statement on the second meeting of the International Health Regulations (2005) Emergency Committee regarding the outbreak of novel coronavirus (2019-nCoV). Retrieved March 28, 2020 from https://www.who.int/news-room/detail/30-01-2020-statement-on-the-second-meeting-of-the-international-health-regulations-(2005)-emergency-committee-regarding-the-outbreak-of-novel-coronavirus-(2019-ncov)
Matthay MA, Aldrich JM, Gotts JE. Treatment for severe acute respiratory distress syndrome from COVID-19. Lancet Respir Med 2020. DOI: 10.1016/S2213-2600(20)30127-2. 18. DOI: https://doi.org/10.1016/S2213-2600(20)30127-2
Kalil AC. Treating COVID-19-off-label drug use, compassionate use, and randomized clinical trials during pandemics. JAMA 2020. DOI: 10.1001/jama.2020.4742. DOI: https://doi.org/10.1001/jama.2020.4742
Gautret P, Lagier J-C. Parolam P et al. Clinical and Microbiological Effect of a Combination Of Hydroxychloroquine And Azithromycin In 80 Covid-19 Patients With At Least A Six-Day Follow Up: A Pilot Observational Study. Travel Medicine and Infectious Disease (2020), doi: https://doi.org/10.1016/j.tmaid.2020.101663. DOI: https://doi.org/10.1016/j.tmaid.2020.101663
Pirnay G, Dantier B, Tourid Wet al. Beneficial effect of the hydroxychloroquine/azithromycin combination in elderly patients with COVID-19: Results of an observational study; Le Pharmacien Hospitalier & Clinicien. 2020. DOI : 10.1016/j.phclin.2020.06.001
Vidal Dictionary (2019). 95th edition, Vidal edition, France
Wang Y, Zhang D, Du G, et al. Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial. Lancet 2020; 395: 1569–78 DOI: https://doi.org/10.1016/S0140-6736(20)31022-9
Leneva IA, Russell RJ, Boriskin YS, et al. “Characteristics of arbidol-resistant mutants of influenza virus: implications for the mechanism of anti-influenza action of arbidol”. Antiviral Research. 2009; 81 (2): 132–40. doi:10.1016/j.antiviral.2008.10.009. PMID 19028526. DOI: https://doi.org/10.1016/j.antiviral.2008.10.009
Pécheur EI, Borisevich V, Halfmann P, et al. “The Synthetic Antiviral Drug Arbidol Inhibits Globally Prevalent Pathogenic Viruses”. Journal of Virology. 2016; 90 (6): 3086–92. doi:10.1128/JVI.02077-15. PMC 4810626. PMID 26739045. DOI: https://doi.org/10.1128/JVI.02077-15
Boriskin YS, Pécheur EI, Polyak SJ. “Arbidol: a broad-spectrum antiviral that inhibits acute and chronic HCV infection”. Virology Journal. 2006; 3: 56. doi:10.1186/1743-422X-3-56. PMC 1559594. PMID 16854226. DOI: https://doi.org/10.1186/1743-422X-3-56
Shi L, Xiong H, He J, et al. “Antiviral activity of arbidol against influenza A virus, respiratory syncytial virus, rhinovirus, coxsackie virus and adenovirus in vitro and in vivo”. Archives of Virology. 2007; 152 (8): 1447–55. doi:10.1007/s00705-007-0974-5. PMID 17497238. DOI: https://doi.org/10.1007/s00705-007-0974-5
Glushkov RG, Gus’kova TA, Krylova LIu, et al. “[Mechanisms of arbidole’s immunomodulating action]”. Vestnik Rossiiskoi Akademii Meditsinskikh Nauk (in Russian) 1999; (3): 36–40. PMID 10222830.
Furuta Y, Takahashi K, Shiraki K, et al.“T-705 (favipiravir) and related compounds: Novel broad-spectrum inhibitors of RNA viral infections”. Antiviral Research. 2009; 82 (3): 95–102. doi:10.1016/j.antiviral.2009.02.198. PMID 19428599. DOI: https://doi.org/10.1016/j.antiviral.2009.02.198
Furuta Y, Gowen BB, Takahashi K,. “Favipiravir (T-705), a novel viral RNA polymerase inhibitor”. Antiviral Research. 2013; 100 (2): 446–54. doi:10.1016/j.antiviral.2013.09.015. PMC 3880838. PMID 24084488. DOI: https://doi.org/10.1016/j.antiviral.2013.09.015
Caroline AL, Powell DS, Bethel LM, et al. “Broad spectrum antiviral activity of favipiravir (T-705): protection from highly lethal inhalational Rift Valley Fever”. PLoS Neglected Tropical Diseases. 2014; 8 (4): e2790. doi:10.1371/journal.pntd.0002790. PMC 3983105. PMID 24722586. DOI: https://doi.org/10.1371/journal.pntd.0002790
Yamada K, Noguchi K, Komeno T, et al. “Efficacy of Favipiravir (T-705) in Rabies Postexposure Prophylaxis”. The Journal of Infectious Diseases. 2016; 213 (8): 1253–61. doi:10.1093/infdis/jiv586. PMC 4799667. PMID 26655300. DOI: https://doi.org/10.1093/infdis/jiv586
Murphy J, Sifri CD, Pruitt R, et al. “Human Rabies – Virginia, 2017”. MMWR. Morbidity and Mortality Weekly Report. 2019; 67(5152): 1410–1414. doi:10.15585/mmwr.mm675152a2. PMC 6334827. PMID 30605446. DOI: https://doi.org/10.15585/mmwr.mm675152a2
Khamitov RA, Loginova S, Shchukina VN, et al. Antiviral activity of arbidol and its derivatives against the pathogen of severe acute respiratory syndrome in the cell cultures. Vopr Virusol. 2008; 53:9–13.
Boriskin YS, Leneva IA, Pecheur EI, et al. a broad-spectrum antiviral compound that blocks viral fusion. Curr Med Chem. 2008; 15:997–1005. doi: 10.2174/092986708784049658. DOI: https://doi.org/10.2174/092986708784049658
Blaising J, Polyak SJ, Pecheur EI. Arbidol as a broad-spectrum antiviral: an update. Antiviral Res. 2014; 107:84–94. doi: 10.1016/j.antiviral.2014.04.006. DOI: https://doi.org/10.1016/j.antiviral.2014.04.006
Gagarinova VM, Ignat'eva GS, Sinitskaia LV, et al. The new chemical preparation arbidol: its prophylactic efficacy during influenza epidemics. Zhural Mikrobiologii Epidemiologii I Immunobiologii. 1993; 5:40‐3.
Hennigan S, Kavanaugh A. Interleukin-6 inhibitors in the treatment of rheumatoid arthritis. Ther Clin Risk Manag. 2008; 4: 767-775 DOI: https://doi.org/10.2147/TCRM.S3470
Swerdlow DI, Holmes MV, Kuchenbaecker KB, et al. The interleukin-6 receptor as a target for prevention of coronary heart disease: a mendelian randomisation analysis. Lancet. 2012; 379: 1214-1224. DOI: https://doi.org/10.1016/S0140-6736(12)60110-X
Kaur S, Bansal Y, Kumar R, et al. A panoramic review of IL‐6: structure, pathophysiological roles and inhibitors. Bioorg Med Chem. 2020; 28 (5):115327. DOI: https://doi.org/10.1016/j.bmc.2020.115327
Stone JH, Tuckwell K, Dimonaco S, et al. Trial of tocilizumab in giant-cell arteritis. N Engl J Med. 2017; 377: 317-328. DOI: https://doi.org/10.1056/NEJMoa1613849
Kotch C, Barrett D, Teachey DT. Tocilizumab for the treatment of chimeric antigen receptor T cell-induced cytokine release syndrome. Expert Rev Clin Immunol. 2019; 15: 813-822 DOI: https://doi.org/10.1080/1744666X.2019.1629904
Antonetti F, Finocchiaro O, Mascia M, et al. A Comparison of the Biologic Activity of Two Recombinant IFN-beta Preparations Used in the Treatment of Relapsing-Remitting Multiple Sclerosis. J. Interferon Cytokine Res. 2002; 22(12):1181–1184. DOI: https://doi.org/10.1089/10799900260475696
Markowitz Clyde E. Interferon-Beta: Mechanism of Action and Dosing Issues. Neurology 2007; 68(24 Suppl 4):S8-11. DOI: https://doi.org/10.1212/01.wnl.0000277703.74115.d2
Food and Drug Administration. 2012. Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/103950s5136lbl.pdf. Accessed April 8, 2020.
Shakoory B, Carcillo JA, Chatham WW, et al. Interleukin-1 receptor blockade is associated with reduced mortality in sepsis patients with features of macrophage activation syndrome: reanalysis of a prior Phase III trial. Crit Care Med. 2016; 44(2):275-281. Available at: https://www.ncbi.nlm.nih.gov/pubmed/26584195. DOI: https://doi.org/10.1097/CCM.0000000000001402
Hirvikoski T, Nordenström A, Lindholm T, et al. Cognitive Functions in Children at Risk for Congenital Adrenal Hyperplasia Treated Prenatally with Dexamethasone. The Journal of Clinical Endocrinology & Metabolism. 2007; 92, 2 : 542–548. DOI: https://doi.org/10.1210/jc.2006-1340
World Health Organization (b) welcomes preliminary results about dexamethasone use in treating critically ill COVID-19 patients on 16 Jun 2020. Welcomes preliminary results about dexamethasone use in treating critically ill COVID-19 patients. Anakinra (Kineret) Prescribing Information.
Piechotta V, Chai KL, Valk SJ, et al. Convalescent plasma or hyperimmune immunoglobulin for people with COVID-19: a living systematic review. Cochrane Database of Systematic Reviews 2020; Issue 7. Art. No.: CD013600. DOI: 10.1002/14651858.CD013600.pub2. DOI: https://doi.org/10.1002/14651858.CD013600.pub2
Savarino A, Boelaert JR, Cassone A et al. Effects of chloroquine on viral infections: an old drug against today's diseases. The Lancet Infectious Diseases. 2003; 3 (11): 722-727. DOI: https://doi.org/10.1016/S1473-3099(03)00806-5
Jung H, Bobba R, Su J, et al. The protective effect of antimalarial drugs on thrombovascular events in systemic lupus erythematosus. Arthritis & Rheumatism. 2010; (62), 3: 863–868 DOI 10.1002/art.27289. DOI: https://doi.org/10.1002/art.27289
Marmor MF, Kellner U, Lai TY, et al. American Academy of OphthalmologyRecommendations on Screening for Chloroquine and Hydroxychloroquine Retinopathy. Ophthalmology. 2016; 123 (6): pp. 1386-1394, 10.1016/j.ophtha.2016.01.058. DOI: https://doi.org/10.1016/j.ophtha.2016.01.058
Yao X, Ye F, Zhang M, et al. In Vitro Antiviral Activity and Projection of Optimized Dosing Design of Hydroxychloroquine for the Treatment of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). Clin Infect Dis. 2020; 10.1093/cid/ciaa237. DOI: https://doi.org/10.1093/cid/ciaa237
Tran DH, Ugamata R, Hirose T, et al. Azithromycin, a 15-membered macrolide antibiotic, inhibits influenza A(H1N1)pdm09 virus infection by interfering with virus internalization process. The Journal of Antibiotics. 2019; 72: 759–768. DOI: https://doi.org/10.1038/s41429-019-0204-x
Le Bideau AMJ, Duflot I, Jardot P, et al. In vitro testing of combined hydroxychloroquine and azithromycin on SARS-CoV-2 shows synergistic effect. Microbial Pathogenesis.2020 ; 145 : 104228 DOI: https://doi.org/10.1016/j.micpath.2020.104228
Rosenberg ES, Dufort EM, Udo T, et al. Association of treatment with hydroxychloroquine or azithromycin with in-hospital mortality in patients with COVID-19 in New York State. JAMA. 2020; 323(24):2493-2502. doi:10.1001/jama.2020.8630. DOI: https://doi.org/10.1001/jama.2020.8630
Mehra MR, Desai S, Ruschitzka F, et al. Hydroxychloroquine or chloroquine with or without a macrolide for treatment of COVID-19: a multinational registry analysis. 2020; https://doi.org/10.1016/ S0140-6736(20)31180-6 DOI: https://doi.org/10.1016/S0140-6736(20)31180-6
Beigel JH, Tomashek KM, Dodd LE, et al. Remdesivir for the Treatment of Covid-19 — Preliminary Report; The New England Journal of Medicine, 2020; DOI: 10.1056/NEJMoa2007764. DOI: https://doi.org/10.1056/NEJMoa2007764
Cao B, Wang Y, Wen D, et al. A Trial of Lopinavir–Ritonavir in Adults Hospitalized with Severe Covid-19, The new england journal of medicine, 2020; 382 no. 19 DOI: https://doi.org/10.1056/NEJMc2008043
Lian N, Xie H, Lin S, et al. Umifenovir treatment is not associated with improved outcomes in patients with coronavirus disease 2019: a retrospective study. Clinical Microbiology and Infectious Diseases. 2020; DOI:https://doi.org/10.1016/j.cmi.2020.04.026. DOI: https://doi.org/10.1016/j.cmi.2020.04.026
Li G, De Clercq E. Therapeutic options for the 2019 novel coronavirus (2019-nCoV). Nature Reviews Drug Discovery. 2020 Feb doi: 10.1038/d41573-020-00016-0. DOI: https://doi.org/10.1038/d41573-020-00016-0
Chunguang Y, Chunjin K, Daoyuan Y, et al. Effectiveness of Arbidol for COVID-19 Prevention in Health. Professionals Front. Public Health. 2020; https://doi.org/10.3389/fpubh.2020.00249. DOI: https://doi.org/10.3389/fpubh.2020.00249
Luo P, Liu Y, Qiu L, et al. Tocilizumab treatment in COVID‐19: A single center experience. J Med Virol . 2020; 10.1002/jmv.25801. DOI: https://doi.org/10.1002/jmv.25801
Xu X, Han M, Li T, et al. Effective treatment of severe COVID-19 patients with tocilizumab. PNAS May 19, 2020; 117 (20) 10970-10975 DOI: https://doi.org/10.1073/pnas.2005615117
Guaraldi G, Meschiari M, Cozzi-Lepri A, et al. Tocilizumab in patients with severe COVID-19: a retrospective cohort study. Lancet Rheumatol. 2020; https://doi.org/10.1016/ S2665-9913(20)30173-9.
Hung IF, Lung KC, Tso EY, et al. Triple combination of interferon beta-1b, lopinavir-ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID-19: an open-label, randomised, phase 2 trials. Lancet. 2020; 395(10238):1695-1704. Available at: https://www.ncbi.nlm.nih.gov/pubmed/32401715.
Davoudi-Monfared E, Rahmani H, Khalili H, et al. Efficacy and Safety of Interferon Β-1a In Treatment of Severe COVID-19: A randomized clinical trial. medRxiv preprint. 2020; doi: https://doi.org/10.1101/2020.05.28.20116467. DOI: https://doi.org/10.1101/2020.05.28.20116467
Zhou Q, Wei X, Xiang X, et al. Interferon-a2b treatment for COVID-19. medRxiv. 2020; Preprint. Available at: https://www.medrxiv.org/content/10.1101/2020.04.06.20042580v1.
Horby P, Lim WS, Emberson J, and the, RECOVERY Collaborative Group. Effect of dexamethasone in hospitalized patients with COVID-19: Preliminary report. medRxiv. 2020 Jun 22.
Huet T, Beaussier H, Voisin O, et al. Anakinra for severe forms of COVID-19: a cohort study. Lancet Rheumatol 2020; 2 (7): e393–400. DOI: https://doi.org/10.1016/S2665-9913(20)30164-8
Dimopoulos G, Mast Q, Markou N, et al. Favorable Anakinra Responses in Severe Covid-19 Patients with Secondary Hemophagocytic Lymphohistiocytosis. Clinical and Translational Report. 2020; 28 (1) : 117-123.e1 DOI: https://doi.org/10.1016/j.chom.2020.05.007
Joyner MJ, Wright RS, Fairweather D, et al. Early safety indicators of COVID-19 convalescent plasma in 5,000 patients. J Clin Invest. 2020; Available at: https://www.ncbi.nlm.nih.gov/pubmed/32525844.
Zeng QL, Yu ZJ, Gou JJ, et al. Effect of Convalescent Plasma Therapy on Viral Shedding and Survival in Patients with Coronavirus Disease 2019. The Journal of Infectious Diseases. 2020; 22 (1) 1: 38–43, https://doi.org/10.1093/infdis/jiaa228. DOI: https://doi.org/10.1093/infdis/jiaa228
Amin I, Shafquat R, Sabina K, et al. Convalescent plasma therapy in the treatment of COVID-19: Practical considerations: Correspondence. International Journal of Surgery. 2020; 79: 204-205. DOI: https://doi.org/10.1016/j.ijsu.2020.05.079
World Health Organisation (c). Clinical management of COVID-19. Interim guidance. WHO; 2020. Available from: https://www.who.int/publications/i/item/clinical-management-ofcovid-19.
Zhu FC, Li YH, Guan XH, et al. Safety, tolerability, and immunogenicity of a recombinant adenovirus type-5 vectored COVID-19 vaccine: a dose-escalation, open-label, non-randomised, first-in-human trial. Lancet. 2020 May 22. DOI: https://doi.org/10.1016/S0140-6736(20)31208-3