Biological control by Plant Growth Promoting Rhizobacteria
Main Article Content
Abstract
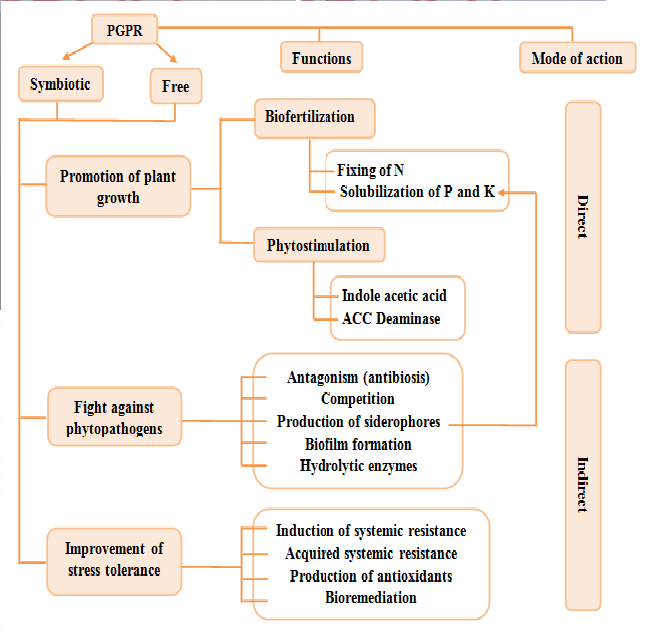
Plant Growth Promoter Rhizobacteria (PGPR) is soil bacteria that can live on, in or around plant tissue and promote plant growth by many mechanisms that include a biological control of plant pathogens. Indeed, PGPRs have a protective effect through several modes of action such as antagonism, competition, production of hydrolytic enzymes and biofilm formation. Moreover, the use of PGPRs as biocontrol agents is very harmonious with the environment and therefore represents a good alternative to the use of chemicals in agriculture. This review is presented as a general bibliographical synthesis on the different aspects of PGPRs and their biocontrol potential.
Article Details
References
Alderman S.C., Coats D.D. and Crowe F.J. Impact of ergot on Kentucky bluegrass grown for seed in northeastern Oregon. Plant Dis. 1996, 80, 853-855. DOI: https://doi.org/10.1094/PD-80-0853
Altieri M.A. The ecological role of biodiversity in agroecosystems. Agri. Ecosystems Environ. 1999, 74, 19-31. DOI: https://doi.org/10.1016/B978-0-444-50019-9.50005-4
Amellal N., Burtin G., Bartoli F., and Heulin T. Colonization of wheat roots by an exopolysaccharide‐producing Pantoea agglomerans strain and its effect on rhizosphere soil aggregation. Appl Environ Microbiol. 1998, 64, 3740–3747. DOI: https://doi.org/10.1128/AEM.64.10.3740-3747.1998
Aouar L. Isolement et identification des actinomycètes antagonistes des microorganismes phytopathogénes. Diplôme de Doctorat en Sciences En Biochimie et Microbiologie Appliquées. Université M’entourai-Constantine, Algérie ; 2012, p35.
Benaissa A. Aspects physiologiques et rhizosphériques de Rhus tripartita (Ucria) Grande en relation avec l’aridité. « Physiological and rhizospheric aspects of Rhus tripartita (Ucria) Grande in relation to aridity”. Doctoral dissertation, university of science and technology of Houari Boumediene, Algiers, Algeria; 2020, p65.
Benaissa A. Plant Growth Promoting Rhizobacteria. A Review, Algerian J. Env. Sc. Technology. 2019, 5(1), 873-880
Benaissa, A., Djebbar, R and Abderrahmani, A. Antagonistic effect of plant growth promoting rhizobacteria associated with Rhus tripartitus on Gram positive and negative bacteria. Analele Universităţii din Oradea, Fascicula Biologie Original Paper Tom. XXVI. 2019, (2), 67-72.
Benbrook C.M., Groth E., Halloran J.M., Hansen M.K. and Marquardt S. Pest management at the crossroads. Consumers union’s of United States Inc. Yonkers. USA; 1996, p272.
Campell R. and Greaves M.P. Anatomy and community structure of the rhizosphere. In the rhizosphere, eds. J. M. Lynch. Wiley Series in Ecological and Applied Microbiologiy, UK; 1990, p11-34
Chauhan P.S and Nautiyal C.S. The purB gene controls rhizosphere colonization by Pantoea agglomerans, Letters in Applied Microbiology. 2010, 50, 205–210. DOI: https://doi.org/10.1111/j.1472-765X.2009.02779.x
Cherif H. Amélioration de la croissance du blé dur en milieu salin par inoculation avec Bacillus sp. et Pantoea agglomerans isolées de sols arides. Thèse de doctorat en sciences. Université Ferhat Abbas Sétif 1, Algérie ; 2014, p177.
Cook R.J. Making greater use of introduced microorganisms for biological control of plant pathogens. Annu. Rev. Phytopathol. 1993, 31, 53–80. DOI: https://doi.org/10.1146/annurev.py.31.090193.000413
Curl E.A and B., Truelove. The rhizosphere. Journal of nutrition and soil science. Springer-Verlag, Berlin-Heidelberg-New York. 1986, p288.
De Kouassi M. Les possibilités de la lutte microbiologique. , VertigO - la revue électronique en sciences de l'environnement [En ligne]. 2001, 2(2). DOI : https://doi.org/10.4000/vertigo.4091 DOI: https://doi.org/10.4000/vertigo.4091
De T. K., Sarkar T. K., De M., Maity T.K. Mukherjee A., Das S. Abundance and occurrence of phosphate solubilizing bacteria and phosphatase in Sediment of Hooghly estuary, north east coast of Bay of Bengal, India. Journal of Coastal Development. 2011, 15(1), 9-16.
Elad Y and Kapat A. The role of Trichoderma harzianum protease in the biocontrol of Botrytis cinerea. European Journal of Plant Pathology. 1999. 105(2), 177-189. DOI: https://doi.org/10.1023/A:1008753629207
Emmert E.A.B. and Handelsman J. Biocontrol of plant disease: a (Gram-) positive perspective. FEMS. Microbiol. Lett. 1999, 171, 1-9. DOI: https://doi.org/10.1111/j.1574-6968.1999.tb13405.x
Errakhi R. Contribution d’actinomycètes (Actinobactéries) à la lutte biologique contre Sclerotium rolfsii et rôle de l’acide oxalique dans l’induction des mécanismes de défense. Thèse de Doctorat. Université Cadi Ayyad, Marrakech Maroc ; 2008.
Fan B., Chen X. H., Budiharjo A., Bleiss W., Vater J. and Borriss R. Efficient colonizationof plant roots by the plant growth promoting bacterium Bacillus amyloliquefaciens FZB42, engineered to express green florescent protein, J of Biotechnol. 2011, 151(4), 303–311. https://doi.org/10.1016/j.jbiotec.2010.12.022 DOI: https://doi.org/10.1016/j.jbiotec.2010.12.022
Fravel D.R. Commercialization and implementation of biocontrol. Ann. Rev. Phytopathol. 2005, 43, 337-359. https://doi.org/10.1146/annurev.phyto.43.032904.092924 DOI: https://doi.org/10.1146/annurev.phyto.43.032904.092924
Goswami D., Thakker J.N., Dhandhukia P.C. Portraying mechanics of plant growth promoting rhizobacteria (PGPR): A review. Cogent Food Agric. 2016, 2(1). https://doi.org/10.1080/23311932.2015.1127500 DOI: https://doi.org/10.1080/23311932.2015.1127500
Gupta, G., Snehi, S. K., Singh, V. Role of PGPR in biofilm formations and its importance in plant health. Biofilms in Plant and Soil Health. 2017, p27-42 DOI: https://doi.org/10.1002/9781119246329.ch2
Iniguez A.L., Dong Y and Triplett E.W. Nitrogen fixation in wheat provided by Klebsiella pneumoniae 342, Mol Plant‐Microbe Interact. 2004, 17(10), 1078–1085. https://doi.org/10.1094/MPMI.2004.17.10.1078 DOI: https://doi.org/10.1094/MPMI.2004.17.10.1078
Ishii H. Impact of fungicide resistance in plant pathogens on crop disease control and agricultural environment. Japan Agricultural Research Quarterly. 2006, 40(3), 205-211. https://doi.org/10.6090/jarq.40.205 DOI: https://doi.org/10.6090/jarq.40.205
Jan A.T., Azam M., Ali A., Rizwanul-Haq Q.M . Novel approaches of beneficial Pseudomonas in mitigation of plant diseases – an appraisal. J Plant Int. 2011, 6(4), 195-205. https://doi.org/10.1080/17429145.2010.541944 DOI: https://doi.org/10.1080/17429145.2010.541944
Jijakly M.H. La lutte biologique en phytopathologie, In : Phytopathology. Lepoivre. Bruxelles, Belgique. 2003, http://hdl.handle.net/2268/37379
Karnwal, A., Production of indole acetic acid by fluorescent Pseudomonas in the presence of L-Tryptophan and rice root exudates. J. Plant Pathol. 2009. 91(1), 61-63. https: // www. jstor .org / stable / 41998574
Kasim W.A., Gaafar R.M., Abou-Ali R.M., Omar, M.N., Hewait H.M. Effect of biofilm forming plant growth promoting rhizobacteria on salinity tolerance in barley. Ann. Agric. Sci. 2016. 61(2), 217–227. https://doi.org/10.1016/j.aoas.2016.07.003 DOI: https://doi.org/10.1016/j.aoas.2016.07.003
Kempf, H. J and Wolf, G. Erwinia herbicola as a biocontrol agent of Fusarium culmorum and Puccinia recondita f. sp. tritici on Wheat. Phytopathology. 1989, 79(2), 990-994. DOI: https://doi.org/10.1094/Phyto-79-990
Khakipour N., Khvazi K., Mojallali H, Pazira E ., Asadirahmani H. Production of auxin hormonr by Fluorescent pseudomonas. American-Eurasian Journal Agriculture end Environment Science 2008, 4(6), 687-692
Kim C., Kecskes M.L., Deaker R.J., Gilchrist K., New P.B and Kennedy I.R. Wheat root colonization and nitrogenase activity by Azospirillum isolates from crop plants in Korea, Can J Microbiol. 2005, 51(11), 948–956. https://doi.org/10.1139/w05-052 DOI: https://doi.org/10.1139/w05-052
Kim J., Rees D.C. Nitrogenase and biological nitrogen fixation. Biochemistry. 1994, 33(2), 389-97. https://doi.org/10.1021/bi00168a001 DOI: https://doi.org/10.1021/bi00168a001
Kloepper J W. Plant-growth-promoting rhizobacteria as biological control agents, In: Soil Microbial Ecology, F.B. Jr., Metting .Marcel Dekker inc., N.Y; 1993, p255-273.
Kloepper, J. W. and Schroth M. N. Plant growth promoting rhizobacteria on radishes. In Proceedings of the 4th International Conference on Plant Pathogenic Bacteriaed. Station and Pathologic Vegetal and Phytobacteriologic. 1978, 2, 879-882.
Kloepper J. W., R. Rodriguez-Ubana G. W. Zehnder J. F. Murphy E. Sikora, and C. Fernandez. Plant root-bacterial interactions in biological control of soilborne diseases andpotential extension to systemic and foliar diseases. Austral. Plant Pathol. 1999, 28, 21–26. https://doi.org/10.1071/AP99003 DOI: https://doi.org/10.1071/AP99003
Kokalis-Burell N., Kloepper J.W., Reddy MS. Plant growth-promoting rhizobacteria as transplant amendments and their effects on indigenous rhizosphere microorganisms. Appl Soil Eco. 2005, 31(1-2), 91-100. https://doi.org/10.1016/j.apsoil.2005.03.007 DOI: https://doi.org/10.1016/j.apsoil.2005.03.007
Krysciak D., Schmeisser C., Preuss S., Riethausen J., Quitschau M., Grond S. Involvement of multiple loci in quorum quenching of autoinducer I molecules in the nitrogen‐fixing symbiont Rhizobium (Sinorhizobium) sp. strain NGR234, Appl Environ Microbiol. 2011, 77(15), 5089–5099. http://dx.doi.org/10.1128/AEM.00112-11. DOI: https://doi.org/10.1128/AEM.00112-11
Leong J. Siderophores: their biochemistry and possible role in the biocontrol of plant pathogens. Annu Rev Phytopathol. 1986, 24, 187-208. https://doi.org/10.1146/annurev.py.24.090186.001155 DOI: https://doi.org/10.1146/annurev.py.24.090186.001155
Lepoivre P. Phytopathologie: bases moléculaires et biologiques des pathosystèmes etfondements des stratégies de lutte. De BoeckUniversité, Bruxelles, Belgium. 2003, p432.
Loper, J. E and Schroth, M. N. Influence of bacterial sources of indole-3-acetic acid on root elongation of sugar beet. Phytopathology. 1986, 76 (4),386-389. DOI: https://doi.org/10.1094/Phyto-76-386
Mao W., Lewis A., Lumsden R.D. and Hebbar K.P. Biocontrol of selected soilborne diseases of tomato and pepper plants. Crop Prot. 1998, 17(6), 535-542. https://doi.org/10.1016/S0261-2194(98)00055-6 DOI: https://doi.org/10.1016/S0261-2194(98)00055-6
Maurhofer M., Reimmann C., Schmidli-Sacherer P., Heeb S., Haas D and Defago G. Salicylic acid biosynthetic genes expressed in Pseudomonas fluorescens strain P3 improve the induction of systemic resistance in tobacco against tobacco necrosis virus. Phytopathology. 1998, 88(7), 678-684. https://doi.org/10.1094/PHYTO.1998.88.7.678 DOI: https://doi.org/10.1094/PHYTO.1998.88.7.678
Mccarthy A.J., Williams, S.T. Actinomycetes as agents of biodegradation in the environment. Gene. 1992, 115(1-2), 189-192. https://doi.org/10.1016/0378-1119(92)90558-7 DOI: https://doi.org/10.1016/0378-1119(92)90558-7
MOUAS BOURBIA, Sophia. Biodisponibilité du potassium dans la rhizosphère d’Olea europaea L. Thèse de doctorat. Thèse doctorat-Université Mouloud Mammeri-Tizi Ouzou. Algérie ; 2013, p19-20.
Nadeem S.M., Naveed M., Zahir Z.A., Asghar H.N. Plant–Microbe Interactions for Sustainable Agriculture: Fundamentals and Recent Advances. In: Arora N. (eds) Plant Microbe Symbiosis: Fundamentals and Advances. Springer, New Delhi; 2013, p51-103. https://doi.org/10.1007/978-81-322-1287-4_2 DOI: https://doi.org/10.1007/978-81-322-1287-4_2
Nadeem, S.M., Zahir, Z.A., Naveed, M and Arshad, M. Preliminary investigations on inducing salt tolerance in maize through inoculation with rhizobacteria containing ACC deaminase activity. Can. J. Microbiol. 2007. 53(10): 1141–1149. https://doi.org/10.1139/W07-081 DOI: https://doi.org/10.1139/W07-081
Nguyen-the C., Carlin F. The microbiology of minimally processed fresh fruits and vegetables. Crit. Rev. Food Sci. Nutr. 1994, 34(4), 371–401. https://doi.org/10.1080/10408399409527668 DOI: https://doi.org/10.1080/10408399409527668
Piano S., Neyrotti V., Migheli Q., Gullino M.L. Biocontrol capability of Metschnikowia pulcherrima against Botrytis postharvest rot of apple. Postharvest Biol. Technol. 1997, 11 (3), 131-140. https://doi.org/10.1016/S0925-5214(97)00022-7 DOI: https://doi.org/10.1016/S0925-5214(97)00022-7
Pieterse C. M. J., Van Wees S.C . M., Hoffland E., Van Pelt J.A., Van Loon L. C. Systemic resistance in Arabidopsis induced by biocontrole bacteria is independent of salycilic acid accumulation and pathogenesis- related gene expression. The Plant Cell. 1996, 8, 1225- 1237. DOI: https://doi.org/10.1105/tpc.8.8.1225 DOI: https://doi.org/10.1105/tpc.8.8.1225
Podile AR and Kishore KG. Plant growth promoting rhizobacteria. In: Gnanamanickam SS (Ed.). Plant-Associated Bacteria, Springer, Dordrecht; 2006, p195–230. DOI: https://doi.org/10.1007/978-1-4020-4538-7_6
Prasanna R., Pattnaik S., Sugitha T.C., Nain L and Saxena A.K. Development of cyanobacterium‐based biofilms and their in vitro evaluation for agriculturally useful traits, Folia Microbiol (Praha). 2011, 56, 49–58. https://doi.org/10.1007/s12223-011-0013-5 DOI: https://doi.org/10.1007/s12223-011-0013-5
Raafat D., Sahl H. G. Chitosan and its antimicrobial potential - a critical literature survey. Microb. Biotechnol. 2009, 2(2), 186–201. https://doi.org/10.1111/j.1751-7915.2008.00080.x DOI: https://doi.org/10.1111/j.1751-7915.2008.00080.x
Raj S. V., Raja A.K., Vimalanathan A.B., Tyagi M.G., Shah N.H., Johnson Amala Justin N.A., Santhose B.I., Sathiyaseelan K. Study of starch degrading bacteria from kitchen waste soil in the production of Amylase by using paddy straw. Recent Research in Science and Technology. 2009, 1(1), 008–013.
Sabaratnam S. and Traquair J.A. Formulation of a Streptomyces biocontrol agent for the suppression of Rhizoctonia damping-off in tomato transplants. Biol. Control. 2002, 23(3), 245-253. https://doi.org/10.1006/bcon.2001.1014 DOI: https://doi.org/10.1006/bcon.2001.1014
Shameer S., Prasad T.N.V.K.V. Plant growth promoting rhizobacteria for sustainable agricultural practices with special reference to biotic and abiotic stresses. Plant Growth Regul. 2018, 84, 603–615. https://doi.org/10.1007/s10725-017-0365-1 DOI: https://doi.org/10.1007/s10725-017-0365-1
Singh A., Mehta S., Singh H.B. and Nautiyal C.S. Biocontrol of collar rot disease of betelvine (Piper betle L.) caused by Sclerotium rolfsii by using rhizosphere-competent Pseudomonas fluorescens NBRI-N6 and P. fluorescens NBRI-N. Cur. Microbiol. 2003, 47, 153-158. https://doi.org/10.1007/s00284-002-3938-8 DOI: https://doi.org/10.1007/s00284-002-3938-8
Souza, R. de, Ambrosini, A., and Passaglia, L.M.P. Plant growth-promoting bacteria as inoculants in agricultural soils. Genet. Mol. Biol. 2015. 38(4): 401–419. https://doi.org/10.1590/S1415-475738420150053 DOI: https://doi.org/10.1590/S1415-475738420150053
Thakore Y., The biopesticide market for global agricultural use, industrial biotechnology. 2006, 2(3), 294-208. https://doi.org/10.1089/ind.2006.2.194 DOI: https://doi.org/10.1089/ind.2006.2.194
Van Loon, L. C., Bakker, P and Pieterse, C. M. J. Systemic resistance induced by rhizosphere bacteria. Annual Review of Phytopathology. 1998, 36, 453- 483. https://doi.org/10.1146/annurev.phyto.36.1.453 DOI: https://doi.org/10.1146/annurev.phyto.36.1.453
Van Nieuwenhove C., Holm V., Kulasooriya S.A and Vlassak K. Establishment of Azorhizobium caulinodans in the rhizosphere of wetland rice (Oryza sativa L.), Biol Fertil Soils. 2000, 31, 143–149. https://doi.org/10.1007/s003740050637 DOI: https://doi.org/10.1007/s003740050637
Vanderlinde E.M., Muszynski A., Harrison J.J., Koval S.F., Foreman D.L and Ceri H., Rhizobium leguminosarum biovar viciae 3841, deficient in 27‐hydroxyoctacosanoatemodified lipopolysaccharide, is impaired in desiccation tolerance, biofilm formation and motility, Microbiol. 2009. 155, 3055–3069. doi: 10.1099/mic.0.025031-0 DOI: https://doi.org/10.1099/mic.0.025031-0
Vinodkumar S, Nakkeeran S, Renukadevi P and Mohankumar S. Diversity and antiviral potential of rhizospheric and endophytic Bacillus species and phyto-antiviral principles against tobacco streak virus in cotton. Agriculture, Ecosystems & Environment. 2018, 267, 42-51. https://doi.org/10.1016/j.agee.2018.08.008 DOI: https://doi.org/10.1016/j.agee.2018.08.008
Weyens N., S. Monchy, S., Vangronsveld. J., Taghavi, S and Vander Lelie., D. PlantMicrobe Partnerships. In K.N. Timmis (ed.), Handbook of hydrocarbon and lipid microbiology, Springer-Verlag, Berlin Heidelberg; 2010, p254-256. DOI: https://doi.org/10.1007/978-3-540-77587-4_189
Williams A., Wilkinson M., Krehenbrink D.M., Russo A., Zorreguieta and J.A Downie, Glucomannan‐mediated attachment of Rhizobium leguminosarum to pea root hairs is required for competitive nodule infection, J Bacteriol. 2008, 190(13), 4706–4715. DOI: 10.1128/JB.01694-07 DOI: https://doi.org/10.1128/JB.01694-07
Yamaga F., Washio K., Morikawa M. Sustainable biodegradation of phenol by Acinetobacter calcoaceticus P23 isolated from the rhizosphere of duckweed Lemna aoukikusa, Environ Sci Technol. 2010, 44 (16), 6470–6474. https://doi.org/10.1021/es1007017 DOI: https://doi.org/10.1021/es1007017
Yang J., Kharbanda P.D and Mirza M. Evaluation of Paenibacillus polymyxa PKB1 for biocontrol of Pythium disease of cucumber in a hydroponic system, Acta Hortic (ISHS). 2004, 635, 59–6. DOI: https://doi.org/10.17660/ActaHortic.2004.635.7
Yang J., Kloepper J.W and Ryu C.M. Rhizosphere bacteria help plants tolerate abiotic stress. Trends in Plant Science. 2009, 14 (1), 1-4. https://doi.org/10.1016/j.tplants.2008.10.004 DOI: https://doi.org/10.1016/j.tplants.2008.10.004
Yang Z, Liu S, Zheng D and Feng S. Effects of cadium, zinc and lead on soil enzyme activities. J. Environ. 2006, 18 (6), 1135-1141. https://doi.org/10.1016/S1001-0742(06)60051-X DOI: https://doi.org/10.1016/S1001-0742(06)60051-X
Zahir, Z.A., Munir, A., Asghar, H.N., Shaharoona, B and Arshad, M. Effectiveness of rhizobacteria containing ACC-deaminase for growth promotion of pea (Pisum sativum) under drought conditions. J. Microbiol. Biotechnol. 2008, 18(5), 958–963.